BPC-157 (5mg / 10mg)
Price range: $52.00 through $97.00
Size: 5mg / 10mg
Contents: BPC-157
Form: Lyophilized powder
Purity: >99%
SKU: P-BPC157
FREE Shipping on $200+ orders
Discount per Quantity
| Quantity | 5 - 8 | 9 + |
|---|---|---|
| Discount | 5% | 10% |
| Price | Price range: $49.40 through $92.15 | Price range: $46.80 through $87.30 |
BPC-157 Peptide
The BPC-157 peptide, also known as Pentadecapeptide BPC 157 or Body Protection Compound 157, is a synthetic compound that has been suggested in various studies to assist with healing joint, tendon, and muscle tissue, as well as nerve tissue.
BPC-157 is a peptide composed of 15 amino acids with potential protective properties. As the name suggests, Body Protection Compound (BPC) is an amino acid fragment isolated from gastric juice.(1) BPC-157 is also commonly known as pentadecapeptide due to the 15 amino acids it is comprised of.(1)
Overview
BPC-157 has been steadily researched for its potential in wound healing. Presentation of BPC-157 may stimulate the growth hormone (GH) receptors, thereby inducing similar GH potential. BPC-157 peptide appears to bind with growth hormone receptors, possibly stimulating cell proliferation. This may lead to the development of new tissue composed of collagen and the development of a network of blood vessels in a process also called ‘angiogenesis.’ Consequently, the wound is ‘rebuilt’ and healed faster than the usual rate.(1)
BPC-157 has also been studied in correlation to gastrointestinal function. Serotonin, an enteric neurotransmitter, is localized in the GI tract and GI mucosa. Altered serotonin levels may inhibit gastric acid secretion, affecting gut mucosal function and influencing gastric blood flow.(2) BPC-157 appears to have a particular antidepressant activity, which may counteract serotonin-induced action. The peptide may counteract the 5-HT2A receptors, restricting the serotonin binding with these receptors and thereby inhibiting its action.(3) The peptide has been researched for its potential action across diverse functions, including tissue repair, pain perception, gastrointestinal regulation, and tendon, ligament, muscle, and bone cell reparations.
Multiple studies have since been conducted to understand the full action of the peptide, especially in the area of healing gastrointestinal ulceration, which is elaborated on below. Studies have suggested the peptide may increase the build-up of the blood vessels and induce anti-inflammation potential via improving functional recovery.(4)
Chemical Makeup
Molecular Formula: C62H98N16O22
Molecular Weight: 1419.55 g/mol
Other Known Titles: Body Protection Compound-157
Research and Clinical Studies
BPC-157 Peptide and Wound Healing
In a study, three experimental murine models were used – first with skin tissue wounds, second with colon tissue anastomosis, and third with synthetic sponge implantation. A portion of the murine models were presented with a placebo, whereas others were presented with the BPC 157 peptide. After the study, all models were histologically examined. The researchers reported that the BPC-157 murine models appeared to exhibit higher numbers of collagen, reticulin, and blood vessel development than the ones in the control group.(5)
In a particular study, researchers explored the theory that the peptide BPC-157 might potentially hasten wound healing compared to a control group. This hypothesis was rooted in observing possible improvements in several key areas of wound healing. These included the formation of new granulation tissue, which is critical in the healing process, along with reepithelialization. In this process, new epithelial cells form to replace those damaged by the wound. Additionally, there was an observation of potential improvements in dermal remodeling, a phase where the skin regains strength and elasticity, and collagen deposition, crucial for tissue repair.(6)
The study also suggested that BPC-157 might have enhanced the expression of vascular endothelial growth factor (VEGF) in the injured skin tissues. VEGF is a significant protein that promotes blood vessel growth, vital to healing damaged tissues. The researchers further speculated that the peptide could have influenced umbilical vein endothelial cell proliferation (HUVECs). These cells line the blood vessels and are considered to be integral to forming new blood vessels during wound healing.(6)
Additionally, there was a conjecture about a noticeable increase in the migration of HUVECs. This observation was based on results from wound healing assays, tests designed to measure various aspects of wound healing. The presence of BPC-157 might have led to an increased expression of VEGF-a, a variant of VEGF, and consequently accelerated the formation of vascular tubes in a laboratory setting. Moreover, the study hinted at the possibility that BPC-157 might influence the activity of specific proteins and enzymes involved in cellular signaling pathways. Specifically, it seemed that BPC-157 could regulate the phosphorylation level of extracellular signal-regulated kinases 1 and 2 (ERK1/2). Phosphorylation is a process that activates or deactivates many protein enzymes and is a crucial step in sending signals within cells. The affected enzymes, ERK1/2, along with their downstream targets, including c-Fos, c-Jun, and Egr-1, are believed to play significant roles in cell growth, migration, and angiogenesis, which is the development of new blood vessels.(6)
BPC-157 Peptide and Tendon Healing
An experiment was conducted in the cultured tendon fibroblasts derived from the tendons of murine models. The cultures were divided into two groups; one was the control, whereas the other was presented with the peptide. Following the study, the following was reported:(1)
- The peptide appeared to promote the outgrowth of tendon fibroblasts and tissue healing;
- Even under H2O2 stress, BPC-157 appeared to stimulate apparent cell survival under stress;
- The peptide appeared to promote migration of the tendon fibroblasts;
- BPC-157 reportedly induced increased levels of phosphorylation of both PAK and paxillin, while the total protein level remained unchanged.
Upon analysis, it was suggested that the peptide may impact tendon healing, tendon outgrowth, and cell survival via the F-actin formation and activation of the FAK and paxillin pathways.(1) F-actin formation is considered a key component in the cell's cytoskeleton, providing structure and aiding in cell movement. If BPC-157 enhances F-actin formation, this might indicate an improvement in the cytoskeletal organization and cell motility of tendon fibroblasts, which are essential for the repair and regeneration of tendon tissues. Further into the study, researchers utilized Western blotting, a laboratory method to detect specific proteins in a sample. Through this analysis, they suggested that BPC-157 might activate focal adhesion kinase (FAK) and paxillin, two proteins that play a significant role in cellular processes. The tentative finding was that the phosphorylation levels of FAK and paxillin appeared to increase in the presence of BPC-157. Interestingly, the total amounts of these proteins appeared to have remained unchanged, leading to the speculation that BPC-157's role might be more about activating existing molecules rather than increasing their production. This led to a further hypothesis that BPC-157 might activate the FAK-paxillin pathway. This pathway is considered to promote cell migration and adhesion, especially in tendon fibroblasts. The activation of this pathway could imply that BPC-157 plays a role in enhancing the movement and adherence of these cells, which are key processes in tendon healing and regeneration.
BPC-157 Peptide and Gastrointestinal Healing
A study was conducted to scrutinize the action of BPC-157 peptide against similar angiogenic growth factors such as EGF, FGF, and VEGF. The primary assumptions were that BPC-157 is highly stable, biocompatible, and sufficient to exert action when presented by itself. While the study reported improved healing, only BPC-157 appeared to have exhibited consistent results in all wound types (i.e., chronic and acute) on the esophagus, stomach, duodenum, and lower GI tract. This study suggested the extent of the angiogenic potential of the peptide is apparently very high as it appeared to extend not only on local wounds and ligaments but also on GI wounds and bone healing.(7)
BPC-157 Peptide and Tissue Damage
A study was conducted to understand the extent of the angiogenic potential of the peptide beyond local wounds, ligaments, and GI tract wounds and to study its action on multiple gastrointestinal lesions on the pancreas, liver injuries, heart damage, endothelium damage, and blood pressure. Following the results, scientists suggested that the BPC-157 peptide may induce a network of activities via peptidergic defense systems. There is also a possibility that BPC-157 may play a role in addressing both acute and chronic inflammation, aiding in wound healing, and assisting in the healing of fractures, including cases of pseudoarthrosis. This broad spectrum of potential suggests that BPC-157 could be part of the organism's unique peptidergic defense system.(8)
There are several neurotransmitters and functions considered by scientists to be important, such as dopamine, nitrous oxide, prostaglandin, and other neuron systems. Any over-activity or inhibition of these systems may lead to lesions in different organs. BPC-157, through its defense system, appears to counteract these systems and possibly reverse their over-activation and inhibition. The researchers commented that these might include important systems, ”namely, dopamine-, NO-, prostaglandin-, somatosensory neuron-system,” and more.(8)
BPC-157 Peptide and Muscle Healing
A study was conducted on murine models with injured gastrocnemius muscle complex. These murine models were then presented with methylprednisolone (corticosteroid). These corticosteroid murine models were then divided into two groups: one was presented with BPC-157, and the other was presented with a placebo. Both compounds were presented once in 24 hours and examined on days 1, 2, 4, 7, and 14. Upon examination, it was reported that the corticosteroid appeared to significantly worsen the muscle damage in the murine models. However, BPC-157 appeared to exhibit apparent signs of healing and restoration of the damaged gastrocnemius muscle and restoring functioning ability.(9)
Amphetamine-Induced Hypersensitivity
Laboratory experiments have suggested that the BPC-157 peptide may have the ability to heal multiple different lesions – in the GI tract, liver, pancreas, and others. This trend in lab findings indicated that the peptide had some interaction with the dopamine system. To investigate further, this study presented the BPC-157 peptide in amphetamine (dopamine agonist) murine models. It was observed that BPC-157 appeared to be able to reverse the amphetamine-induced excitability in the murine models. Furthermore, murine models were presented with another dopamine agonist, haloperidol, and then presented with amphetamine on days 1, 2, 4, and 10. These murine models were then presented with BPC-157 to illustrate its action. Upon examination, it was suggested by the researchers that the peptide appeared to cause an almost complete reversal of the haloperidol action.(10)
BPC-157 Peptide and Central Nervous System
In a particular study using a murine model, researchers explored the potential of BPC-157 in the context of traumatic brain injury (TBI). BPC-157 might have played a role in significantly reducing the damage caused by TBI in experimental models, as indicated by improved early outcomes in the experiments conducted. During the critical 24-hour period following the injury, the observations hinted a minimal mortality rate in the BPC-157 group. Furthermore, the severity of traumatic lesions typically associated with TBI, such as subarachnoid hemorrhage (bleeding in the space between the brain and the tissues that cover it), intraventricular hemorrhage (bleeding inside the brain's ventricular system), brain laceration, and hemorrhagic laceration, appeared to be less pronounced in the murine models of the BPC-157 group. This suggested a protective potential of the peptide against such injuries.(11)
Another interesting observation was the considerable improvement in brain edema, swelling in the brain tissue often caused by traumatic injuries. The hypothesis extended to the possibility that if BPC-157 were introduced before the occurrence of TBI, it might show an improved ratio of conscious/unconscious/death states in the test subjects. In other words, the peptide might potentially prevent or reduce the severity of unconsciousness and lower mortality rates associated with TBI in experimental models. Moreover, there was a suggestion that the immediate exposure of BPC-157 immediately before the injury may have mitigated the damage in the murine models subjected to a force impulse, typically used to simulate TBI in research. This hinted at the possibility of the peptide having preventive or protective potential against the immediate consequences of traumatic brain injury in experimental models.(11)
BPC 157 peptide is available for research and laboratory purposes only. Please review and adhere to our Terms and Conditions before ordering.
References:
- Chang, Chung-Hsun et al. “The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration.” Journal of applied physiology (Bethesda, Md. : 1985) vol. 110,3 (2011): 774-80. doi:10.1152/japplphysiol.00945.2010. https://pubmed.ncbi.nlm.nih.gov/21030672/
- Ormsbee, H S 3rd, and J D Fondacaro. “Action of serotonin on the gastrointestinal tract.” Proceedings of the Society for Experimental Biology and Medicine. Society for Experimental Biology and Medicine (New York, N.Y.) vol. 178,3 (1985): 333-8. doi:10.3181/00379727-178-42016. https://pubmed.ncbi.nlm.nih.gov/3919396/
- Sikiric, Predrag et al. “Brain-gut Axis and Pentadecapeptide BPC 157: Theoretical and Practical Implications.” Current neuropharmacology vol. 14,8 (2016): 857-865. doi:10.2174/1570159x13666160502153022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5333585/#r1
- Krivic, A., Majerovic, M., Jelic, I. et al. Modulation of early functional recovery of Achilles tendon to bone unit after transection by BPC 157 and methylprednisolone. Inflamm. res. 57, 205–210 (2008). https://doi.org/10.1007/s00011-007-7056-8
- S Seiwerth, et al. “BPC 157's effect on healing.” Journal of physiology, Paris vol. 91,3-5 (1997): 173-8. doi:10.1016/s0928-4257(97)89480-6. https://pubmed.ncbi.nlm.nih.gov/9403790/
- Huang, T., Zhang, K., Sun, L., Xue, X., Zhang, C., Shu, Z., Mu, N., Gu, J., Zhang, W., Wang, Y., Zhang, Y., & Zhang, W. (2015). Body protective compound-157 enhances alkali-burn wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro. Drug design, development and therapy, 9, 2485–2499. https://doi.org/10.2147/DDDT.S82030
- Seiwerth, Sven et al. “BPC 157 and Standard Angiogenic Growth Factors. Gastrointestinal Tract Healing, Lessons from Tendon, Ligament, Muscle and Bone Healing.” Current pharmaceutical design vol. 24,18 (2018): 1972-1989. doi:10.2174/1381612824666180712110447. https://pubmed.ncbi.nlm.nih.gov/29998800/
- Sikiric P. (1999). The pharmacological properties of the novel peptide BPC 157 (PL-10). Inflammopharmacology, 7(1), 1–14. https://doi.org/10.1007/s10787-999-0022-z https://pubmed.ncbi.nlm.nih.gov/17657443/
- Pevec D, Novinscak T, Brcic L, Sipos K, Jukic I, Staresinic M, Mise S, Brcic I, Kolenc D, Klicek R, Banic T, Sever M, Kocijan A, Berkopic L, Radic B, Buljat G, Anic T, Zoricic I, Bojanic I, Seiwerth S, Sikiric P. Impact of pentadecapeptide BPC 157 on muscle healing impaired by systemic corticosteroid application. Med Sci Monit. 2010 Mar;16(3):BR81-88. PMID: 20190676. https://pubmed.ncbi.nlm.nih.gov/20190676/
- Jelovac, N et al. “A novel pentadecapeptide, BPC 157, blocks the stereotypy produced acutely by amphetamine and the development of haloperidol-induced supersensitivity to amphetamine.” Biological psychiatry vol. 43,7 (1998): 511-9. doi:10.1016/s0006-3223(97)00277-1. https://pubmed.ncbi.nlm.nih.gov/9547930/
- Tudor, M., Jandric, I., Marovic, A., Gjurasin, M., Perovic, D., Radic, B., Blagaic, A. B., Kolenc, D., Brcic, L., Zarkovic, K., Seiwerth, S., & Sikiric, P. (2010). Traumatic brain injury in mice and pentadecapeptide BPC 157 effect. Regulatory peptides, 160(1-3), 26–32. https://doi.org/10.1016/j.regpep.2009.11.012
- Gwyer, D., Wragg, N.M. & Wilson, S.L. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell Tissue Res 377, 153–159 (2019). https://doi.org/10.1007/s00441-019-03016-8
- Veljaca, Marija et al, The development of PL 14736 for treatment of inflammatory bowel disease, Advanced in GI pharmacology, 2002 O-32. https://www.bib.irb.hr/192824
- Phase I clinical trial in healthy volunteers to study safety and pharmacokinetics of BPC-157, a pentadecapeptide from gastric source. https://clinicaltrials.gov/ct2/show/NCT02637284?
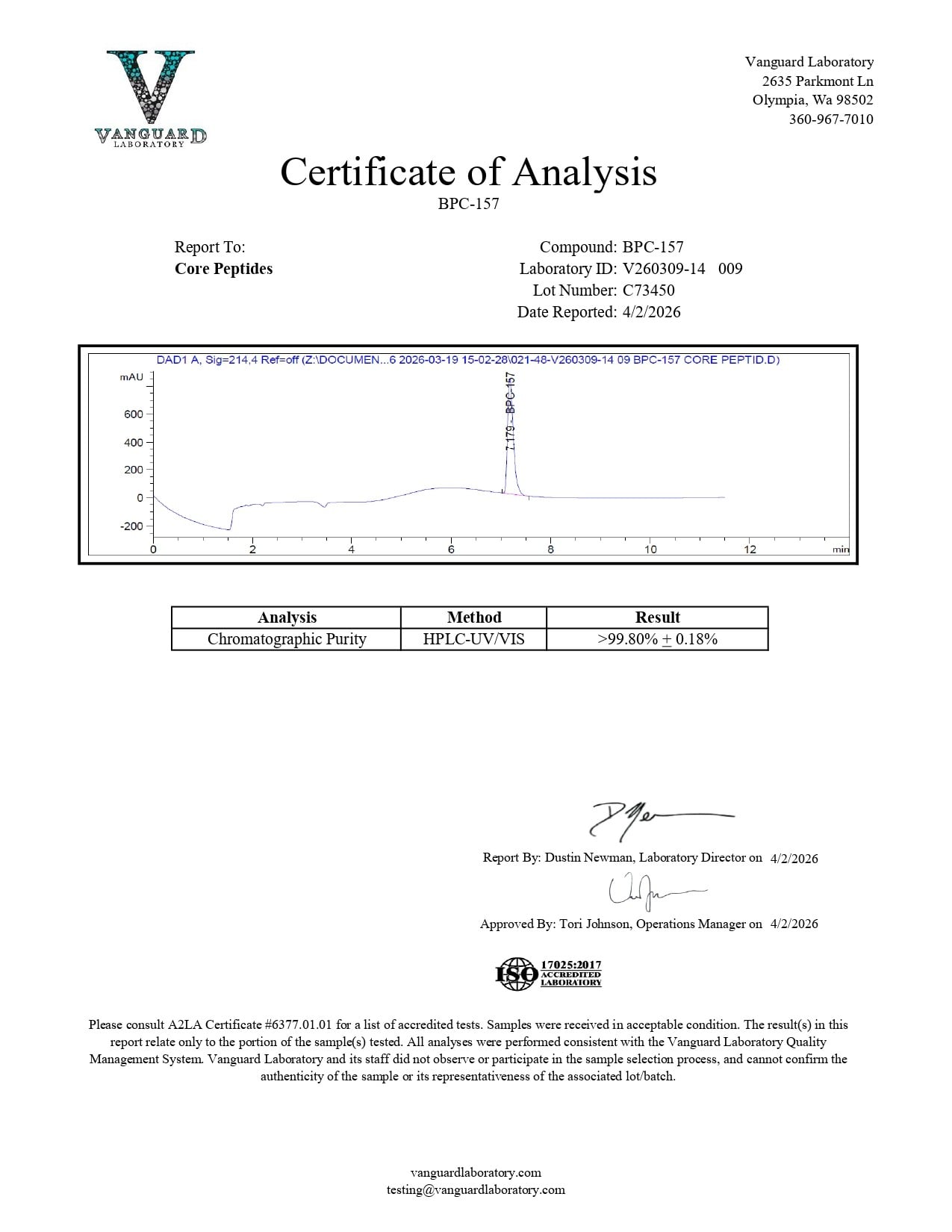
BPC-157 (5mg) – Certificate of Analysis
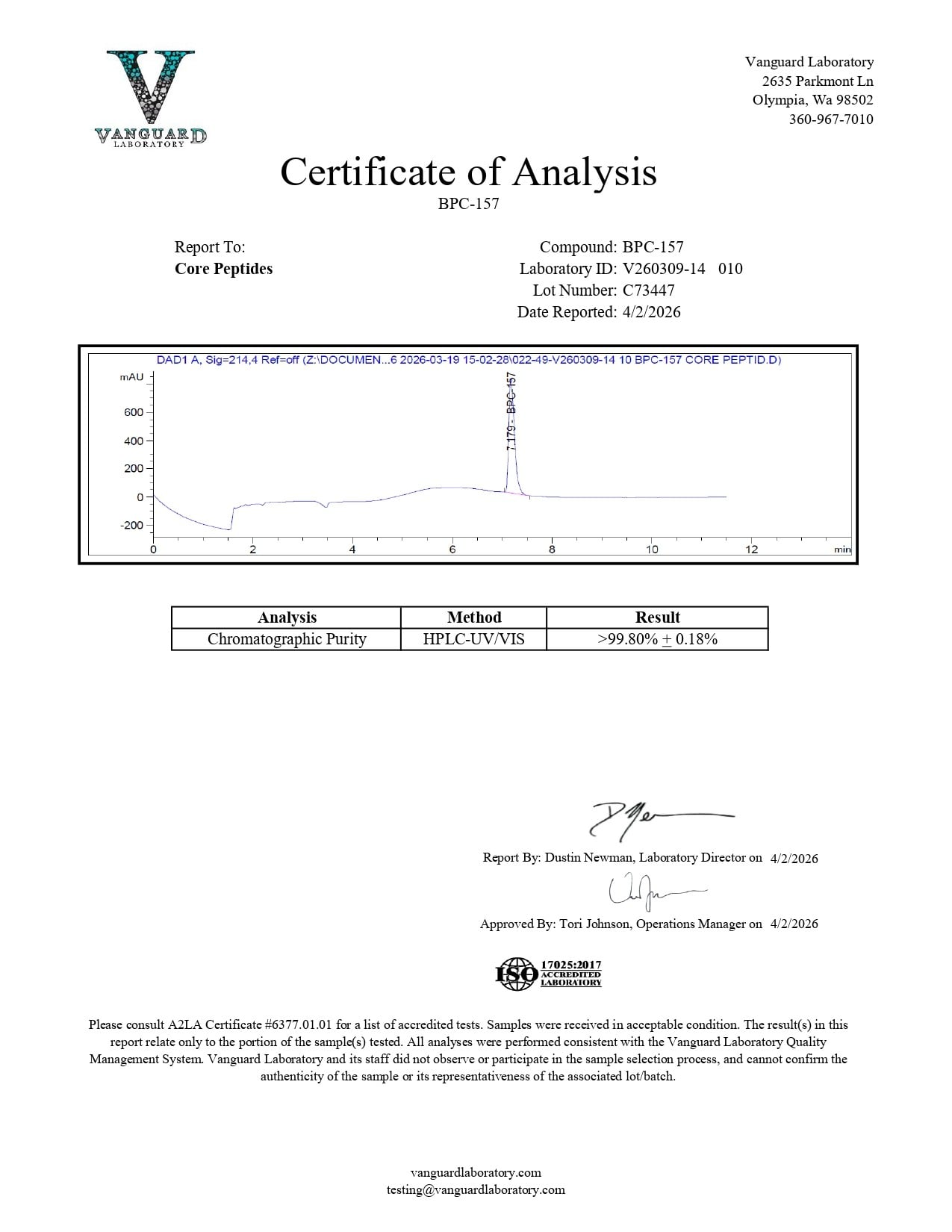
BPC-157 (10mg) – Certificate of Analysis