KPV (4mg)
Original price was: $45.00.$38.00Current price is: $38.00.
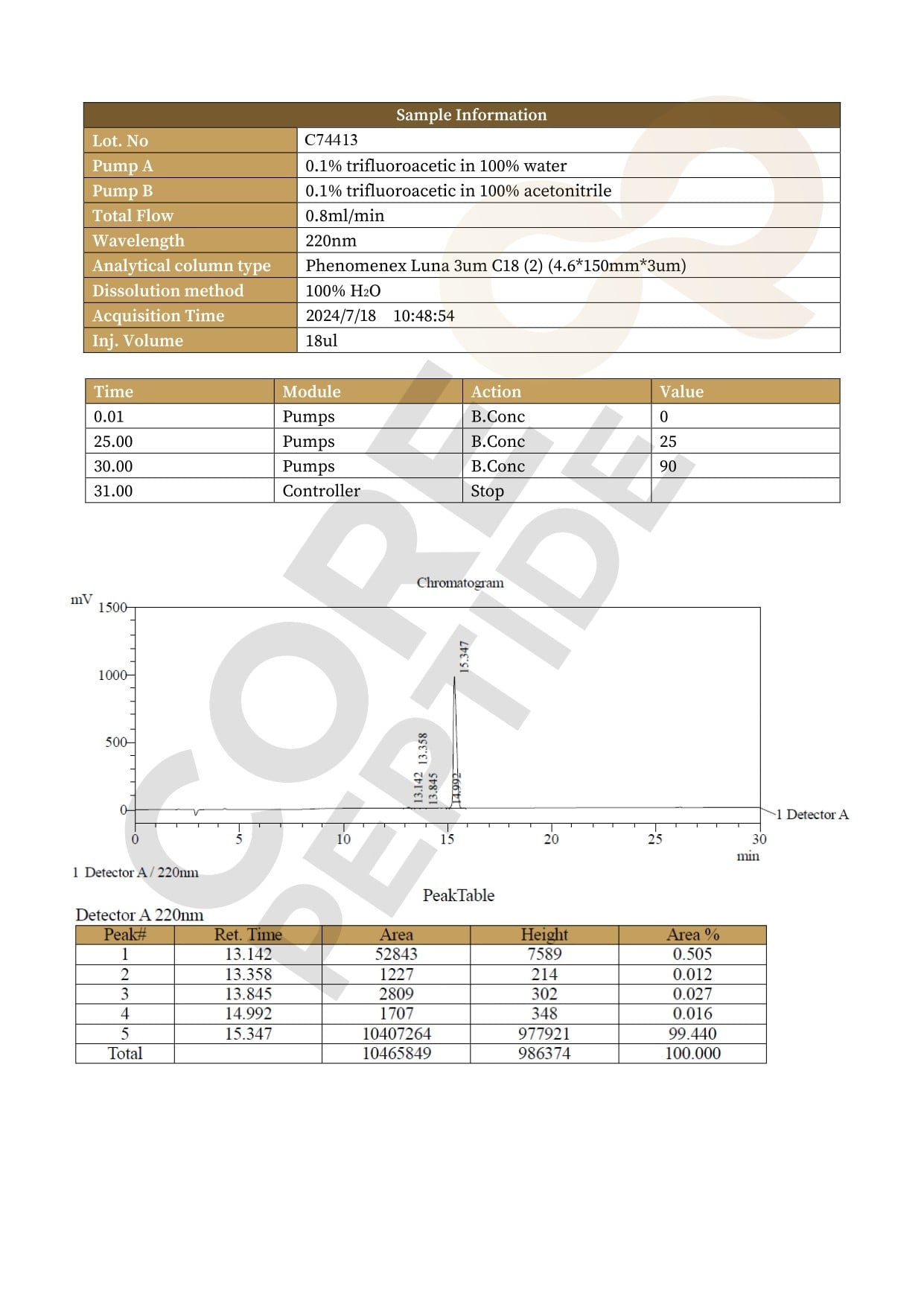
Size: 4mg
Contents: KPV (4mg)
Form: Lyophilized powder
Purity: >99%
SKU: P-KPV-4
FREE Shipping on $200+ orders
Discount per Quantity
| Quantity | 5 - 8 | 9 + |
|---|---|---|
| Discount | 5% | 10% |
| Price | $36.10 | $34.20 |
KPV Peptide
Alpha melanocyte-stimulating hormone (a-MSH) is also classified as a melanotropin. a-MSH is an endogenous peptide hormone, composed of 13 amino acids, and considered to play a role in metabolic function, as well as other biological processes. Scientists isolated a fragment of this protein hormone and identified its potential biological action, naming it KPV peptide. KPV comprises three amino acids: Lysine, Proline, and Valine.(1) This peptide is a C-terminal fragment of the a-MSH protein hormone, which is considered the primary amino acid sequence in the hormone responsible for its properties.(2)
A study(2) was published in 1989 explaining how the tripeptide was isolated and its biological potential determined. Upon discovering that the COOH terminal peptide in the a-MSH hormone is the primary amino acid messenger sequence, scientists conducted preliminary research to determine if KPV might prevent an excessive increase in vasopermeability and excessive swelling of blood vessels. As a part of the study, scientists isolated the KPV peptide and presented it to experimental mice to determine its potential to mitigate swelling in their ears. After the completion of the study, the researchers reported that the isolated fragment appeared to have inhibited the swelling. KPV's potential anti-inflammatory action may be induced by inactivating the inflammatory pathways.(3) It may also possibly inhibit the synthesis and release of the pro-inflammatory cytokine cells in intestinal and immune cells.
Chemical Makeup
Molecular Formula: C16H30N4O4
Molecular Weight: 342.43 g/mol
Other Known Titles: MSH (11-13), ACTH(11-13), alpha-MSH(11-13)
Research and Clinical Studies
KPV Peptide and Intestinal Protection
A study(4) was conducted on murine models to determine the peptide's potential on intestinal inflammation. The experiment was conducted on mice induced with bowel dysfunction. These mice were divided into two groups: one group was given the peptide, and the other was given the placebo. After the study, researchers reported that the peptide mice exhibited reduced inflammatory cells and anti-enzymatic symptoms.
Another study(4) was conducted on a murine model of inflamed intestines, which involved the exposure of a chemical-induced compound of KPV and a chemical called hyaluronic acid. This chemical-induced KPV compound was given to the mice, with the added hyaluronic acid supplementation, intended to aid targeted delivery of the peptide to specific locations in the intestine. The results observed mitigated swelling in the intestine.
KPV Peptide and Intestinal Cells
One study(5) was conducted on a cell culture of inflamed intestinal cells. The main purpose of this study was to determine the peptide’s potential against inflammation. Inflamed intestinal cells were isolated and exposed to either the KPV peptide or a placebo. Upon exposure to the peptide, these cells were examined, and results indicated that even nanomolar concentrations of the peptide appeared to have led to anti-inflammatory results. The researchers suggested that the KPV peptide appeared to mainly act via PepT1 expression in these intestinal cells, suggesting that PepT1 may play a role in transporting the peptide to the site of inflammation.
Another study delved into the potential of KPV in addressing ulcerative inflammation of the colonic mucosa cells.(6) The researchers hypothesized that KPV might mitigate inflammatory responses within colonic cells, by accelerating mucosal healing and alleviating inflammation of the colonic mucosa. The action mechanism was suggested to involve the targeted delivery of KPV to inflamed colonic tissues, where it may exert anti-inflammatory potential. The researchers found that KPV may have exhibited a capacity to protect mucosal surfaces and downregulate TNF-α, a key marker of inflammation.
Further trials in two murine models of intestinal inflammation suggest that KPV may have led to significant improvements, including earlier recovery, significant regain of body weight, and a reduction in inflammatory infiltrates, in the colonic tissue.(7) These outcomes were further supported by a notable decrease in myeloperoxidase (MPO) activity, indicating reduced neutrophil accumulation and inflammation in colonic tissue following KPV exposure. Moreover, the study explored whether KPV's anti-inflammatory actions may have been linked to the melanocortin-1 receptor (MC1R), suggesting that the action of KPV might at least be partially independent of MC1R signaling.
One 1984 study aimed to evaluate the potential antipyretic action of the peptide,(8) wherein rabbits were given KPV peptide to examine its potential action on the nervous system. Following the study, researchers suggested that the peptide exhibited antipyretic potential, reducing the rabbits’ body temperature to optimal levels.
KPV Peptide and Inflammation Studies
A comparative study analysis(9) was conducted to examine the potential of a-MSH and KPV on the swelling (inflammation) of organs. An experiment was conducted on the mice with swollen ears due to skin rashes and dermatitis. The mice were divided into two groups - one was given an irritant (to induce ear swelling) and then exposed to the peptide, and the other with the irritant and the a-MSH molecule. After 24 hours, both groups exhibited apparently equal improvement in reducing ear swelling. After 2 weeks, researchers ceased exposure to both compounds, and only the irritant was given, with the results observing that the a-MSH mice appeared to continue to show reduced swelling compared to the other group.
KPV Peptide and Wound Healing
Wound healing is a complex biological process comprised of three general phases: inflammation, proliferation, and remodeling of the skin, tissue, or cells. This process is characterized by different types of cells and concentrations of cytokines in the wounded area. Though every wound and associated cells affected by the wound may differ, most cells possess a melanocortin 1 receptor (MC1R) receptor. This receptor is where the a-MSH hormone binds, and researchers suggest that a-MSH hormone analogs, such as KPV peptide, may also bind to these receptors.(10)
Another study explored the potential of KPV in enhancing corneal epithelial wound healing with a particular focus on the possible involvement of nitric oxide (NO) in these actions. Following mechanical abrasion to damage the corneal epithelium, the tissue was exposed to variable concentrations of the KPV peptide. The progress of epithelial wound healing was meticulously tracked and analyzed through computerized software, comparing the mean area of the epithelial defect among experimental groups at multiple time intervals. The findings suggested a potential acceleration in the healing process in corneal tissues compared to placebo. Specifically, within 60 hours, all corneas exposed to KPV appeared to have complete re-epithelialization, a stark contrast to the placebo group, where none of the corneas appeared to have achieved full healing.
This accelerated healing effect was apparently hindered by pre-treatment with the nitric oxide synthase inhibitor, Nω-nitro-l-arginine methyl ester (l-NAME), suggesting that the facilitating impact of KPV on corneal epithelial wound healing might be linked to NO activity within the corneal tissue. Further in vitro experiments with corneal epithelial cells (RCE) exposed to different concentrations of KPV reportedly exhibited a stimulation of cell viability at 1 and 10 μM concentrations. These results suggest that KPV may not only accelerate corneal epithelial wound healing but also might stimulate cell viability, hinting at a broader reparative role that may involve NO dynamics.(11)
KPV Peptide and Scar Formation
A study(12) was conducted to further understand the potential of KPV peptide in scar recovery. The experiment was conducted in murine models, half of the group of young mice were exposed to KPV, and the other served as a control group. Half an hour after exposure, these mice underwent two surgical 6.5 mm wide incisions in their dorsal skin under anesthesia. The wound healing and scar formation were analyzed on days 3, 7, 40, and 60. On days 3 and 7, researchers observed that the peptide mice appeared to show improved healing on the skin, possibly due to reduced levels of inflammatory cells such as leukocytes and mast cells. On days 40 and 60, it was observed that the peptide mice exhibited a lesser scar area than the control group.
KPV peptide is available for research and laboratory purposes only. Please review and adhere to our Terms and Conditions before ordering.
References:
- Dalmasso, G., Charrier-Hisamuddin, L., Nguyen, H. T., Yan, Y., Sitaraman, S., & Merlin, D. (2008). PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology, 134(1), 166–178. https://doi.org/10.1053/j.gastro.2007.10.026
- Hiltz ME, Lipton JM. Antiinflammatory activity of a COOH-terminal fragment of the neuropeptide alpha-MSH. FASEB J. 1989 Sep;3(11):2282-4. https://pubmed.ncbi.nlm.nih.gov/2550304/
- Brzoska T, Luger TA, Maaser C, Abels C, Böhm M. Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocr Rev. 2008 Aug;29(5):581-602. doi: 10.1210/er.2007-0027. Epub 2008 Jul 8. https://pubmed.ncbi.nlm.nih.gov/18612139/
- Klaus Kannengiesser, MD, Christian Maaser, MD, Jan Heidemann, MD, Andreas Luegering, MD, Matthias Ross, MD, Thomas Brzoska, PhD, Markus Bohm, MD, Thomas A. Luger, MD, Wolfram Domschke, MD, Torsten Kucharzik, MD, Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease, Inflammatory Bowel Diseases, Volume 14, Issue 3, 1 March 2008, Pages 324–331, https://doi.org/10.1002/ibd.20334
- Dalmasso G, Charrier-Hisamuddin L, Nguyen HT, Yan Y, Sitaraman S, Merlin D. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008 Jan;134(1):166-78. https://pubmed.ncbi.nlm.nih.gov/18061177/
- Xiao, B., Xu, Z., Viennois, E., Zhang, Y., Zhang, Z., Zhang, M., Han, M. K., Kang, Y., & Merlin, D. (2017). Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis. Molecular therapy : the journal of the American Society of Gene Therapy, 25(7), 1628–1640. https://doi.org/10.1016/j.ymthe.2016.11.020
- Kannengiesser K, Maaser C, Heidemann J, Luegering A, Ross M, Brzoska T, Bohm M, Luger TA, Domschke W, Kucharzik T. Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease. Inflamm Bowel Dis. 2008 Mar;14(3):324-31. doi: 10.1002/ibd.20334. PMID: 18092346.
- D.B. Richards, J.M. Lipton, Effect of α-MSH 11–13 (lysine-proline-valine) on fever in the rabbit, Peptides, Volume 5, Issue 4, 1984, Pages 815-817, ISSN 0196-9781, https://doi.org/10.1016/0196-9781(84)90027-5
- Luger, T. A., & Brzoska, T. (2007). alpha-MSH related peptides: a new class of anti-inflammatory and immunomodulating drugs. Annals of the rheumatic diseases, 66 Suppl 3(Suppl 3), iii52–iii55. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2095288/#!po=3.33333
- Brzoska T, Luger TA, Maaser C, Abels C, Böhm M. Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocr Rev. 2008 Aug;29(5):581-602. https://pubmed.ncbi.nlm.nih.gov/18612139/
- Bonfiglio V, Camillieri G, Avitabile T, Leggio GM, Drago F. Effects of the COOH-terminal tripeptide alpha-MSH(11-13) on corneal epithelial wound healing: role of nitric oxide. Exp Eye Res. 2006 Dec;83(6):1366-72. doi: 10.1016/j.exer.2006.07.014. Epub 2006 Sep 11. PMID: 16965771.
- de Souza KS, Cantaruti TA, Azevedo GM Jr, Galdino DA, Rodrigues CM, Costa RA, Vaz NM, Carvalho CR. Improved cutaneous wound healing after intraperitoneal injection of alpha-melanocyte-stimulating hormone. Exp Dermatol. 2015 Mar;24(3):198-203. https://pubmed.ncbi.nlm.nih.gov/25431356/