MOTS-C (10mg)
$116.00
Size: 10mg
Contents: MOTS-C
Form: Lyophilized powder
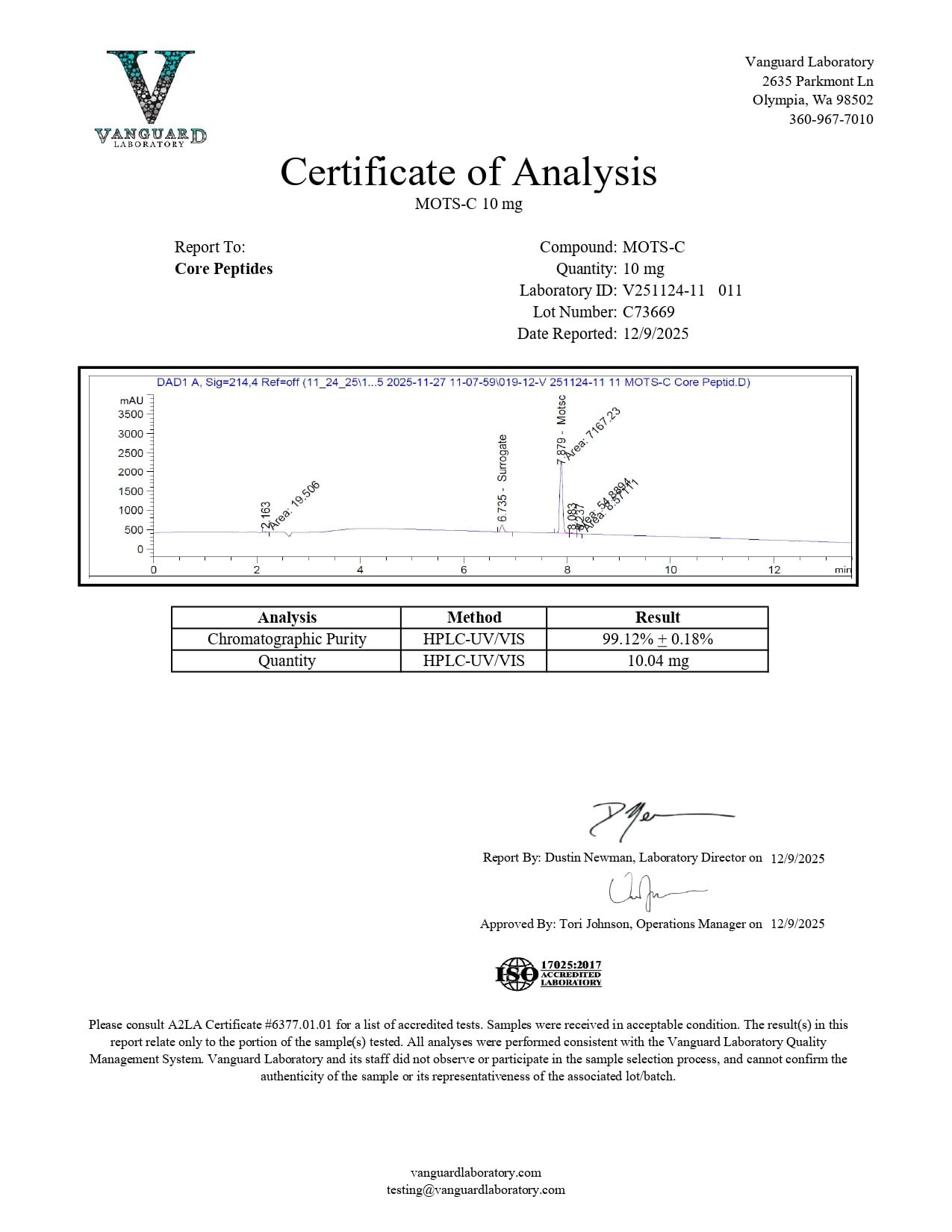
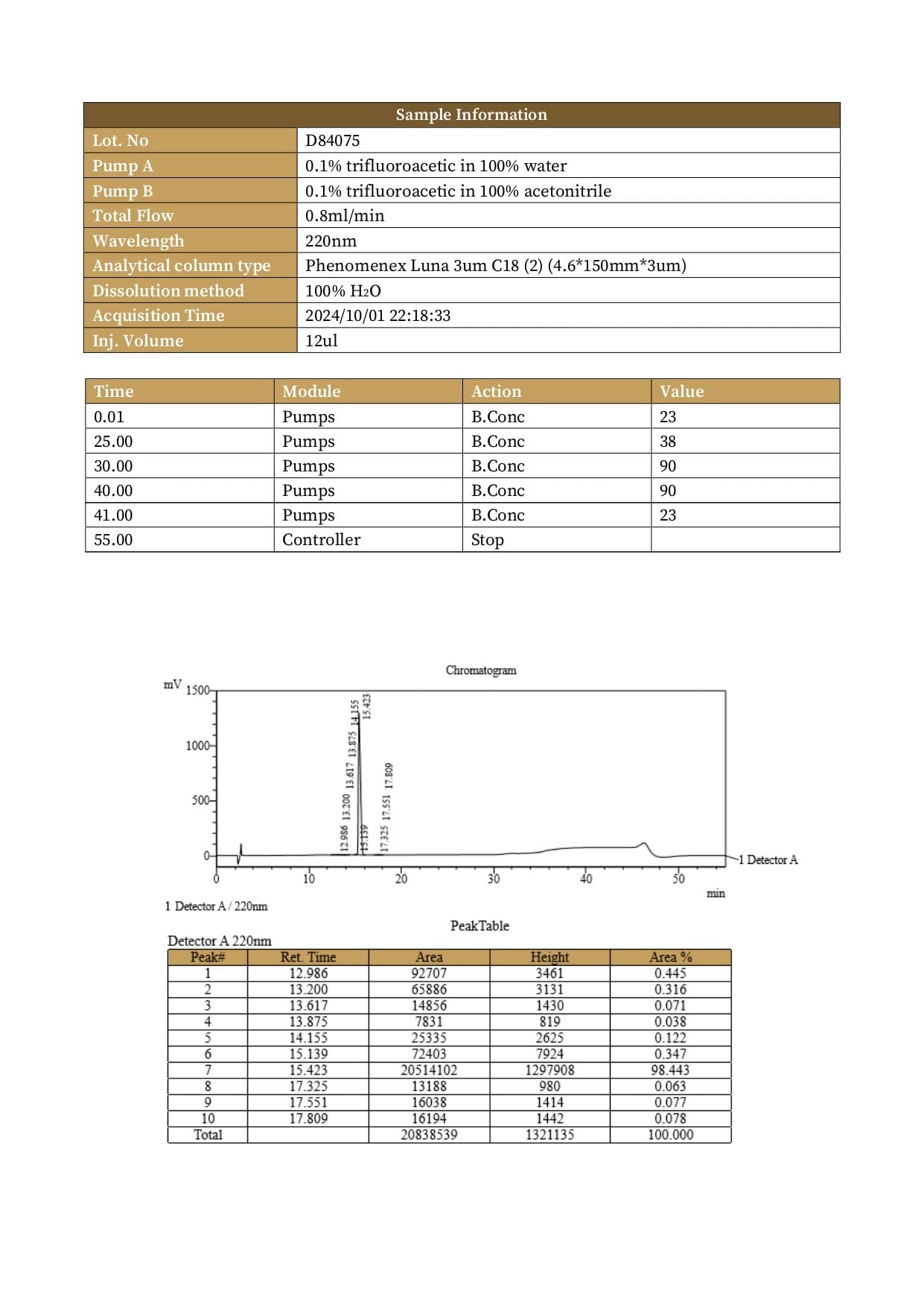
Purity: >99%
SKU: P-MOTS-C-10mg
FREE Shipping on $200+ orders
Discount per Quantity
| Quantity | 5 - 8 | 9 + |
|---|---|---|
| Discount | 5% | 10% |
| Price | $110.20 | $104.40 |
MOTS-c Peptide
MOTS-c (mitochondrial open-reading-frame of the 12S rRNA-c) peptide is a novel mitochondria-derived peptide. It is a short peptide composed of 16 amino acids, expressed in tissues and plasma, indicating a cell-specific and hormonal role.(1) With the potential to work both as a cell-specific compound and as a hormone, this peptide possibly acts by stimulating the AMP-activated protein kinase (AMPK) pathway. Only two mitochondrial-derived peptides (MDPs) have been studied, Humanin and MOTS-c. When metabolic stress occurs in the organism, the peptide is believed to translocate to the cellular nuclei and alter the gene expression. MOTS-c peptide may also be released extracellularly and is known as "mitochondrial hormone" or simply as "mitokine.” (2)(3)
Chemical Makeup(4)
Molecular Formula: C101H152N28O22S2
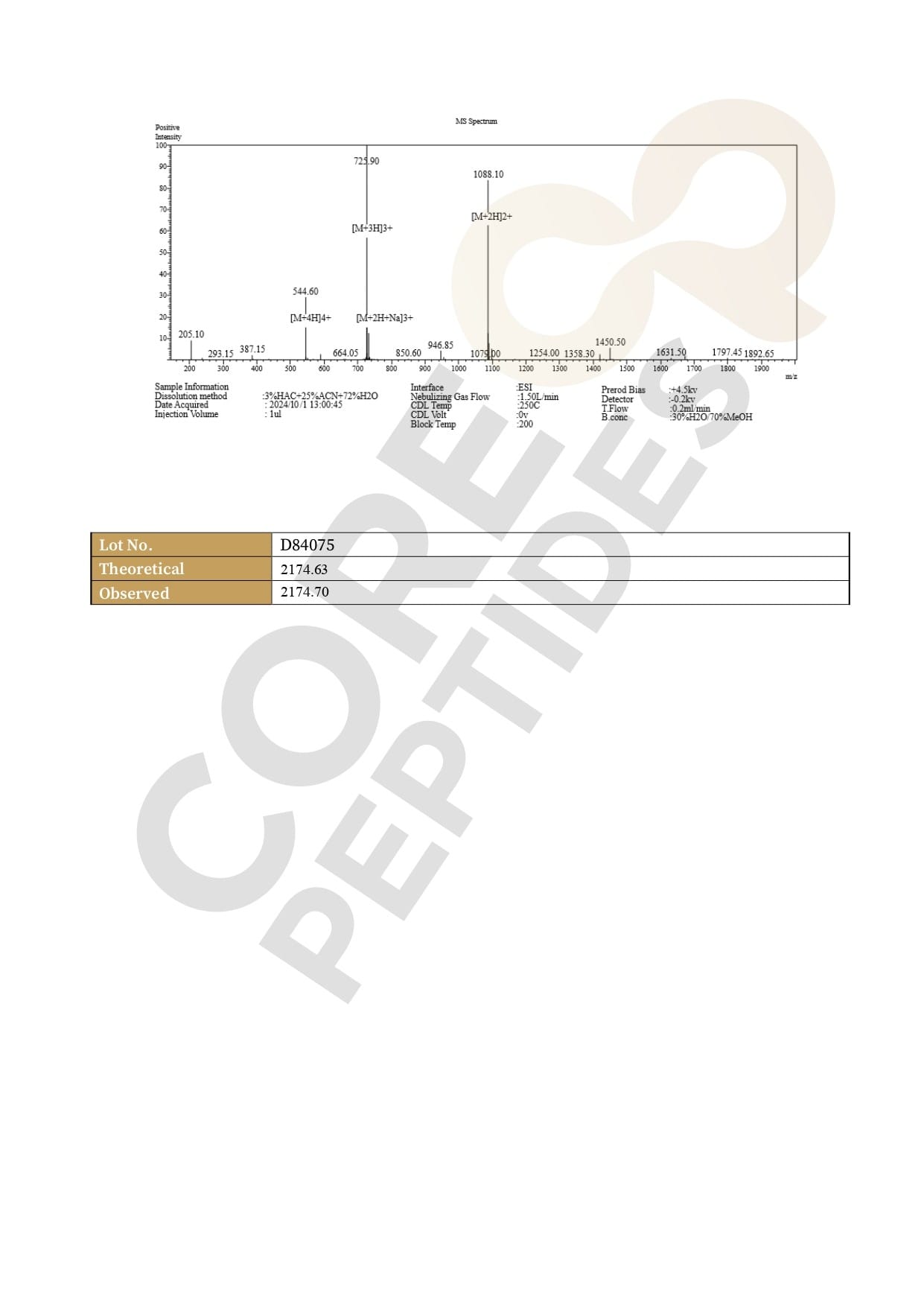
Molecular Weight: 2174.64 g/mol
Other Titles: Mitochondrial-derived peptide MOTS-c, Mitochondrial open reading frame of the 12S rRNA-c
Research and Clinical Studies
Animal research models have indicated multiple potential actions from MOTS-c peptide, including increased physical performance, regulated cellular and tissue metabolism, and myoblast adaptation.(2) Research suggests that these actions may primarily depend on age and age-related changes in MOTS-c expression. The researchers suggest MOTS-c levels and activity might decline, hinting at a role in the cell aging process and the development of age-related metabolic dysfunction. Furthermore, MOTS-c may interact with known aging regulators, such as NAD+ and sirtuins, suggesting its involvement in pathways potentially modulating the lifespan of the cell.(1) As per Joseph C Reynolds et al., “Mitochondria are chief metabolic organelles with strong implications in cell aging that also coordinate broad physiological functions, in part, using peptides that are encoded within their independent genome.”(4) The peptide endogenous expression has also been posited to be boosted via physical activity, potentially enhancing cellular metabolism.(5)
MOTS-c Peptide and Muscle Metabolism
With increasing age, skeletal muscles tend to gain insulin resistance, leading to decreased glucose uptake. Upon peptide exposure, skeletal muscles may be stimulated with an improved response toward AMPK activation. As a result, glucose transporter expression may increase, potentially improving skeletal muscle metabolism and enhancing skeletal muscle functioning and growth. Further, MOTS-c's actions are posited to include targeting metabolic pathways such as the folate-methionine cycle and purine biosynthesis. This targeting may potentially lead to a modulation of cellular metabolism, including actions on glucose uptake and lipid utilization. The peptide's impact might involve a shift in metabolic priorities within the cell, possibly affecting the balance between anabolic and catabolic processes. In systemic metabolism, MOTS-c is posited to function as a mitochondrial hormone, with circulating peptide levels appearing to affect metabolic functions in skeletal muscle and possibly adipose tissue. Its potential regulatory actions on glucose homeostasis and insulin action suggest a broader hormonal role in energy balance and nutrient sensing across different tissues.(1)
MOTS-c Peptide and Fat Cell Metabolism
Research has suggested that the peptide may potentially leave the mitochondrial site, translocate to cellular nuclei, and possibly alter gene expression. More specifically, the peptide may interact with a broad range of genes, particularly those with antioxidant response elements (ARE), hinting at a potential regulatory relationship with stress-responsive transcription factors like NRF2. Such findings suggest a genetically integrated system of mitonuclear communication, where both mitochondrial and nuclear genomes may encode factors that cross-regulate each other. This action, in turn, may alter glucose uptake restriction.(6) This hypothesis was first suggested from a study in which the experimental mice were given high-fat food, and only half were presented with the peptide. The researchers indicated that MOTS-c may potentially impact cellular metabolism by inhibiting the folate cycle directly tethered de novo purine biosynthesis, consequently leading to AMPK activation. Such actions hint at a broader role of the peptide in regulating insulin sensitivity and metabolic homeostasis, offering insights into its preventive potential against age-dependent and high-fat-induced insulin resistance and diet-induced obesity. The study presents supportive data to suggest that the peptide may stimulate glucose utilization, affect the methionine-folate cycle, and promote AMPK activation. These cellular actions suggest that MOTS-c might coordinate various metabolic processes, including glucose and lipid metabolism. Consequently, the murine models exposed to the peptide were lean and more energetic than the rest, further indicating that the peptide might prevent fat accumulation and induce glucose uptake via the AMPK pathway.(3)
MOTS-c Peptide and Bone
MOTS-c peptide has been suggested to regulate the transforming growth factor beta (TGF-beta)/SMAD pathway, which may profoundly affect bone tissues.(7) More specifically, MOTS-c's actions may involve the upregulation of TGF-β/Smad pathway-related genes, including TGF-β1, TGF-β2, and Smad7, suggesting a pivotal role of this pathway in MOTS-c mediated osteogenic differentiation. This hypothesis is further supported when the osteogenic differentiation promoted by MOTS-c is reversed upon TGF-β1 knockdown, indicating that MOTS-c's actions may be at least partly mediated through the TGF-β/Smad pathway. The peptide may also stimulate the expression of osteogenesis-related genes such as ALP, Bglap, and Runx2. Thus, this peptide may stimulate the SMAD pathway in the osteoblast cells, possibly improving bone density and strength. When studied in bone marrow cells, this compound appeared to trigger the differentiation of the stem cells, which may lead to bone tissue development.
MOTS-c Peptide and Cardiac Function
The peptide has not been suggested by researchers to directly influence cardiac function; instead, researchers posit that the peptide exerts potential on the endothelial cells that line the blood vessels inside. These endothelial tissues are considered to affect blood pressure and clotting. The researchers suspect a positive correlation exists between MOTS-c levels and microvascular and epicardial endothelial function. Such findings tentatively suggest MOTS-c as a potential biomarker for endothelial function, with the study revealing a nuanced relationship between MOTS-c levels and vascular reactivity. Further, the research suggested that when mice were exposed to MOTS-c, it appeared to improve the endothelial tissues' functioning, thereby possibly facilitating dysfunction. The mechanistic basis for MOTS-c's action on endothelial function remains speculative but may involve the activation of AMPK.(8)
MOTS-c Peptide and Cell Lifespan
Research has suggested that the peptide may be associated with enhanced longevity on a cellular level. The peptide typically contains glutamate residue, but when this is replaced by lysine, the new compound may exert a functional change. Scientists so far are aware that the functionality of the glutamate and lysine groups are vastly different, but how this specific structural change affects peptide functionality is yet to be understood. Noriyuki Fuku et al. suggests that there is “a biological link between MOTS-c and extended lifespan through the putative endocrine action of this mitokine. Further mechanistic research is needed to determine the functional significance of polymorphism and the potential influence of MOTS-c in the [...] aging process.” (9)
The peptide is available for research and laboratory purposes only. Please review and adhere to our Terms and Conditions before ordering.
References:
- Lee C, Kim KH, Cohen P. MOTS-c: A novel mitochondrial-derived peptide regulating muscle and fat metabolism. Free Radic Biol Med. 2016 Nov;100:182-187. doi: 10.1016/j.freeradbiomed.2016.05.015. Epub 2016 May 20. PMID: 27216708; PMCID: PMC5116416. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5116416/
- Mohtashami Z, Singh MK, Salimiaghdam N, Ozgul M, Kenney MC. Most Recent Mitochondrial Derived Peptide in Human Aging and Age-Related Diseases. Int J Mol Sci. 2022 Oct 9;23(19):11991. doi: 10.3390/ijms231911991. PMID: 36233287; PMCID: PMC9570330.
- Lee C, Zeng J, Drew BG, Sallam T, Martin-Montalvo A, Wan J, Kim SJ, Mehta H, Hevener AL, de Cabo R, Cohen P. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015 Mar 3;21(3):443-54. doi: 10.1016/j.cmet.2015.02.009. PMID: 25738459; PMCID: PMC4350682.
- Lu H, Wei M, Zhai Y, Li Q, Ye Z, Wang L, Luo W, Chen J, Lu Z. MOTS-c peptide regulates adipose homeostasis to prevent ovariectomy-induced metabolic dysfunction. J Mol Med (Berl). 2019 Apr;97(4):473-485. doi: 10.1007/s00109-018-01738-w. Epub 2019 Feb 6. PMID: 30725119. https://pubmed.ncbi.nlm.nih.gov/30725119/
- Reynolds JC, Lai RW, Woodhead JST, Joly JH, Mitchell CJ, Cameron-Smith D, Lu R, Cohen P, Graham NA, Benayoun BA, Merry TL, Lee C. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nat Commun. 2021 Jan 20;12(1):470. https://pubmed.ncbi.nlm.nih.gov/33473109/
- Kim KH, Son JM, Benayoun BA, Lee C. The Mitochondrial-Encoded Peptide MOTS-c Translocates to the Nucleus to Regulate Nuclear Gene Expression in Response to Metabolic Stress. Cell Metab. 2018 Sep 4;28(3):516-524.e7. doi: 10.1016/j.cmet.2018.06.008. Epub 2018 Jul 5. PMID: 29983246; PMCID: PMC6185997. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6185997/
- Hu BT, Chen WZ. MOTS-c improves osteoporosis by promoting osteogenic differentiation of bone marrow mesenchymal stem cells via TGF-β/Smad pathway. Eur Rev Med Pharmacol Sci. 2018 Nov;22(21):7156-7163. doi: 10.26355/eurrev_201811_16247. PMID: 30468456. https://pubmed.ncbi.nlm.nih.gov/30468456/
- Qin Q, Delrio S, Wan J, Jay Widmer R, Cohen P, Lerman LO, Lerman A. Downregulation of circulating MOTS-c levels in patients with coronary endothelial dysfunction. Int J Cardiol. 2018 Mar 1;254:23-27. doi: 10.1016/j.ijcard.2017.12.001. Epub 2017 Dec 6. PMID: 29242099. https://pubmed.ncbi.nlm.nih.gov/29242099/
- Noriyuki Fuku el al., The mitochondrial-derived peptide: A player in exceptional longevity?, http://dx.doi.org/10.1111/acel.12389.